Saito 1905 (syn. Rhizopus microsporus var. oligosporus (Saito) Schipper & Stalpers)
Rhizopus oligosporus is a mold belonging to the family Mucoraceae and is widely used to produce fermented food products. Even though originating from a harmful Rhizopus pathogen species, R. oligosporus is essentially harmful to humans when consumed. Furthermore, the species can produce certain beneficial compounds which limit the growth of potentially harmful microorganisms.
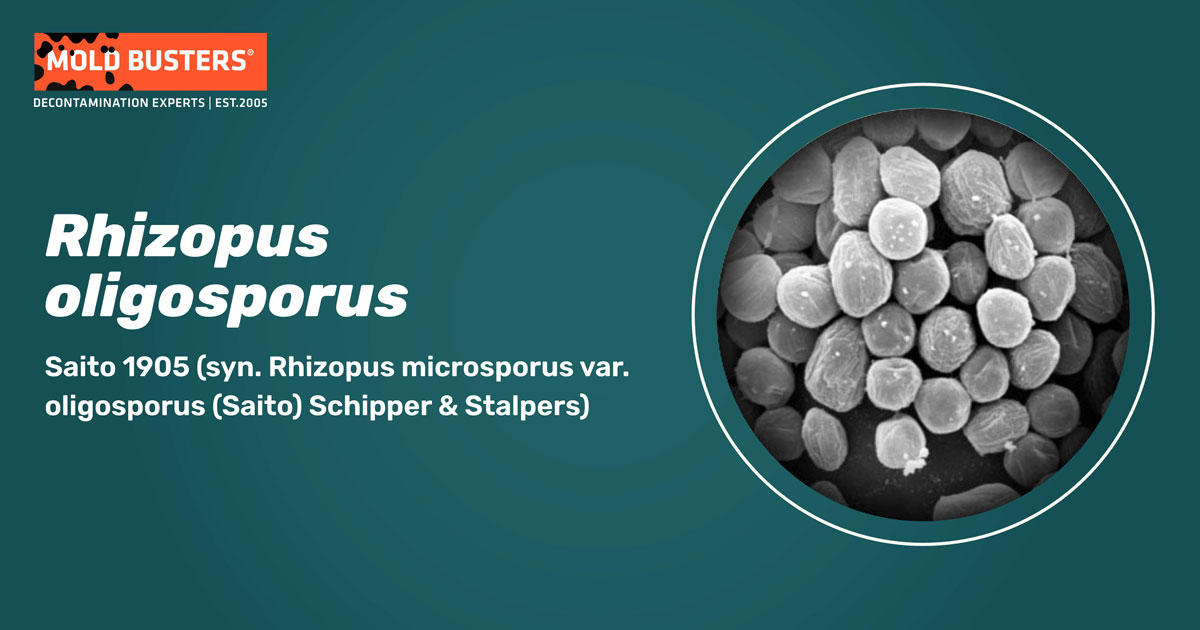
Rhizopus oligosporus classification
Rhizopus oligosporus is today classified as R microsporus var. oligosporus. Several Rhizopus taxa (including R. oligosporus) were considered separate species based on morphology traits such as propagule size, shape, and ornamentation. However, with modern molecular tools, we now distinguish the R. oligosporus as an R. microsporus variety, belonging to the section “micro-Rhizopus” (1). Furthermore, R. oligosporus is considered to be a domesticated form of R. microsporus, which has lost its ability to produce toxins through the process of domestication (2,3).
Rhizopus oligosporus morphology and cultivation
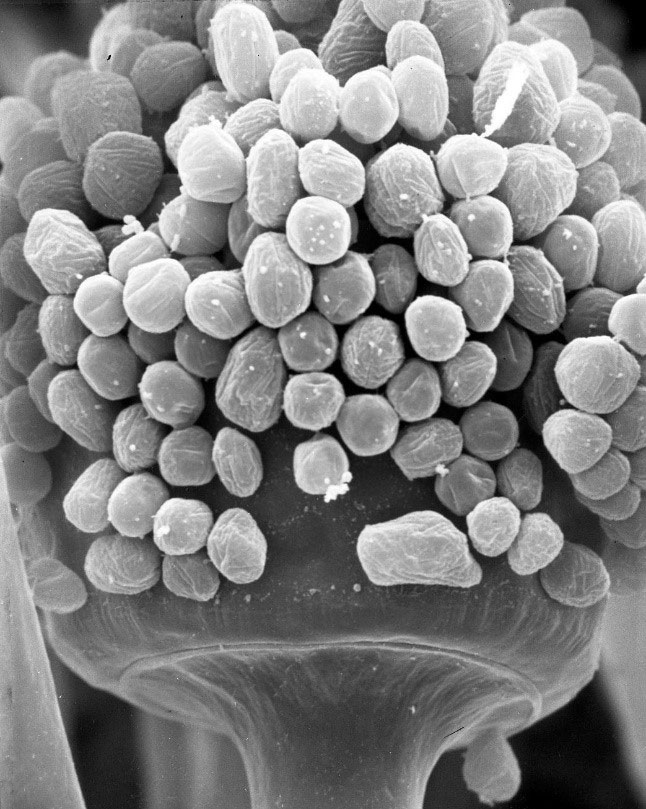
Colonies are pale yellowish to greyish-brown. Rhizoids are simple, semi-translucent (subhyaline). Sporangiophores and rhizoids appear on stolons up to 300 μm long and 15 μm wide, brownish. Sporangiophores emerge in groups of 1-3. Sporangia are dark, up to 80 μm in diameter. Sporangiospores globose to subglobose, mostly up to 9 μm diameter, diverse in size, high proportion irregular spores (>10 %) (Fig. 1) (1,3).
Rhizopus oligosporus optimally grows at temperatures between 30-35 °C (86-95 °F), with a minimum growing temperature of 12 °C (53.6 °F), and a maximum of 45 °C (107.6 °F) (4). The hyphal extension rate is somewhat sensitive to the pH, and the optimum value should be kept at pH= 5.85 (5,6). R. oligosporus grows best at the media water activity of Aw= 0.98 and a 12.5% CO2.
Rhizopus oligosporus fermentation
Pure Rhizopus oligosporus cultures are used to produce Tempeh – a fermented soy-based dish popular in Indonesia. In essence, tempeh is a molded soybean cake, and R. oligosporus is the preferred mold starter for tempeh production for several reasons.
To create tempeh, soybeans must be soaked in water first and kept soaked overnight at a temperature similar to the surrounding environment. Even though air temperature can get rather high in Indonesia, R. oligosporus can grow quite effectively at high temperatures (30-40°C). Under these conditions, R. oligosporus exhibits strong enzymatic activity needed to create the desirable properties in tempeh. Furthermore, the benefits of using R. oligosporus for tempeh production are also the rapid spore germination and allelopathy against other competing microorganisms. It produces certain metabolites (antibiotics and chitinases) which can inhibit the growth of other molds and bacteria, including the potentially toxic Aspergillus flavus and Staphylococcus aureus (3,4).
Tempeh
Principally, the initial stage in tempeh production is to inoculate the soybeans with a starter culture of R. oligosporus, and the beans are then spread into a thin layer. As such, the soybeans are fermented for two to three days at a temperature around 30°C (86°F). During the fermentation process, the pH of the substrate should be maintained at pH= 3-5 by adding a mild acidulant such as vinegar. Next, the mass needs to cool down for optimal and rapid mycelium growth, but it will naturally rise afterward (due to the microorganism action). After around 4 hours, as the fungi growth declines, the beans will be compacted into a solid mass by the fungal mycelium (Fig. 2) (3,4,7).

A more traditional way of producing tempeh is by using the leaves of Hibiscus tiliaceus. As the trichomes growing on the underside of H. tiliaceus leaves naturally may contain R. oligosporus, the soybeans are pressed and stored into the leaves. The resulting fermentation, if not contaminated, should result in whitish tempeh (7,8). On average, 100 g (3.5 oz) of tempeh contains 192 kcal (803 kJ), of which 7.64 g are carbohydrates, 10.80 g are fats, and 20.29 g of proteins. Tempeh also contains various vitamins such as B1 to B3, B6, B9, minerals, and water (9).

Did you know?
The #1 toxic mold type found in kitchens in Canada is the Penicillium/Aspergillus mold group?! Find out more exciting mold stats and facts on our mold statistics page.
Bibliography
- Schipper, M. A. (1984). Revision of the genus Rhizopus. Stud. Mycol., 25, 1-34.
- Jennessen, J., Nielsen, K. F., Houbraken, J., Lyhne, E. K., Schnürer, J., Frisvad, J. C., & Samson, R. A. (2005). Secondary metabolite and mycotoxin production by the Rhizopus microsporus group. Journal of agricultural and food chemistry, 53(5), 1833-1840.
- Jennessen, J., Schnürer, J., Olsson, J., Samson, R. A., & Dijksterhuis, J. (2008). Morphological characteristics of sporangiospores of the tempe fungus Rhizopus oligosporus differentiate it from other taxa of the R. microsporus group. Mycological research, 112(5), 547-563.
- Duniaji, A. S., Wisaniyasa, W., Puspawati N. N, Indri, D. N. (2019) Isolation and Identification of Rhizopus oligosporus Local Isolate Derived from Several Inoculum Sources. IntJCurrMicrobiolAppSci, 8(9):1085-98.
- Sparringa, R. A., Kendall, M., Westby, A., & Owens, J. D. (2002). Effects of temperature, pH, water activity, and CO2 concentration on the growth of Rhizopus oligosporus NRRL 2710. Journal of Applied Microbiology, 92(2), 329-337.
- Wang, H. L., Swain, E. W., & Hesseltine, C. W. (1975). Mass production of Rhizopus oligosporus spores and their application in tempeh fermentation.
- Watanabe, N., Fujimoto, K., & Aoki, H. (2007). Antioxidant activities of the water-soluble fraction in tempeh-like fermented soybean (GABA-tempeh). International Journal of Food Sciences and Nutrition, 58(8), 577-587.
- Muzdalifah, D., Athaillah, Z. A., Nugrahani, W., & Devi, A. F. (2017, January). Colour and pH changes of tempe during extended fermentation. In AIP conference proceedings(Vol. 1803, No. 1, p. 020036). AIP Publishing LLC.
- USDA FoodData Central [Internet]. Available from: fdc.nal.usda.gov

Get Special Gift: Industry-Standard Mold Removal Guidelines
Download the industry-standard guidelines that Mold Busters use in their own mold removal services, including news, tips and special offers:

Written by:
Dusan Sadikovic
Mycologist – MSc, PhD
Mold Busters
Fact checked by:
Michael Golubev
General Manager
Mold Busters
